Porphine
Porphine or porphin is an organic chemical compound with formula C20H14N4. The molecule, which is flat, consists of four pyrrole-like rings joined by four methine (=CH−) groups to form a larger macrocycle ring, which makes it the simplest of the tetrapyrroles. It is classified as an aromatic and heterocyclic compound.[1]
 | |
 | |
| Names | |
|---|---|
| Other names
Porphin | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.002.690 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C20H14N4 | |
| Molar mass | 310.35196 g/mol |
| Appearance | Dark red, shiny leaflets |
| Melting point | N/A |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Porphine is only of theoretical interest. It has been detected in GC-MS of certain fractions of piper betle.[2]
Porphine derivatives: porphyrins
Substituted derivatives of porphine are called porphyrins. Many porphyrins are found in nature with the dominant example being protoporphyrin IX.[3] Many synthetic porphyrins are also known, including octaethylporphyrin[4] and tetraphenylporphyrin.[5]
- Common porphyrins
 Derivatives of protoporphyrin IX are common in nature, the precursor to hemes.
Derivatives of protoporphyrin IX are common in nature, the precursor to hemes. Octaethylporphyrin (H2OEP) is a synthetic analogue of protoporphyrin IX. Unlike the natural porphyrin ligands, OEP2− is highly symmetrical.
Octaethylporphyrin (H2OEP) is a synthetic analogue of protoporphyrin IX. Unlike the natural porphyrin ligands, OEP2− is highly symmetrical. Tetraphenylporphyrin (H2TPP)is another synthetic analogue of protoporphyrin IX. Unlike the natural porphyrin ligands, TPP2− is highly symmetrical. Another difference is that its methine centers are occupied by phenyl groups.
Tetraphenylporphyrin (H2TPP)is another synthetic analogue of protoporphyrin IX. Unlike the natural porphyrin ligands, TPP2− is highly symmetrical. Another difference is that its methine centers are occupied by phenyl groups.
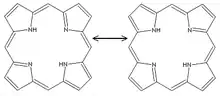
Two resonance structures of porphine.
Further reading
- Budavari, Susan (1989). "7574. Porphine". The Merck Index (11th ed.). Merck & Co., Inc. p. 1210. ISBN 0-911910-28-X. LCCN 89-60001.
References
- "Porphyrin". Encyclopedia of Inorganic and Bioinorganic Chemistry. Wiley-VCH. 2011. doi:10.1002/9781119951438.eibd0638. ISBN 9781119951438.
- Karak S, Das S, Biswas M, Choudhury A, Dutta M, Chaudhury K, De B (December 2019). "Phytochemical composition, β-glucuronidase inhibition, and antioxidant properties of two fractions of Piper betle leaf aqueous extract". Journal of Food Biochemistry. 43 (12): e13048. doi:10.1111/jfbc.13048. PMID 31581322.
- Paul R. Ortiz de Montellano (2008). "Hemes in Biology". Wiley Encyclopedia of Chemical Biology. John Wiley & Sons. doi:10.1002/9780470048672.wecb221. ISBN 978-0470048672.
- Jonathan L. Sessler; Azadeh Mozaffari; Martin R. Johnson (1992). "3,4-Diethylpyrrole and 2,3,7,8,12,13,17,18-Octaethylporphyrin". Org. Synth. 70: 68. doi:10.15227/orgsyn.070.0068.
- Lindsey, Jonathan S. (2000). "Synthesis of meso-substituted porphyrins". In Kadish, Karl M.; Smith, Kevin M.; Guilard, Roger (eds.). Porphyrin Handbook. Vol. 1. pp. 45–118. ISBN 0-12-393200-9.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.